Tertiary wastewater treatment, also known as advanced wastewater treatment, is the third step of wastewater treatment. After secondary treatment, tertiary treatment of effluent entails several extra procedures to minimise organics, turbidity, nitrogen, phosphorus, metals, and pathogens.
Tertiary Wastewater Treatment plays a crucial role in producing high-quality effluent that meets strict environmental standards. Before exploring the top physicochemical treatment methods like reverse osmosis and Electrodialysis, it is important to understand what is tertiary wastewater treatment and why it is essential.
This advanced stage goes beyond primary and secondary processes, removing fine suspended solids, nutrients, pathogens, and dissolved contaminants that conventional systems cannot eliminate. As part of modern tertiary treatment in wastewater, industries and municipalities use filtration, chemical dosing, disinfection, and membrane systems to achieve superior purity.
These processes fall under advanced wastewater treatment processes, designed to protect ecosystems and support water reuse. By learning the types of tertiary wastewater treatment, readers gain a complete picture of how physicochemical techniques ensure cleaner and safer water for diverse applications.
- What is Tertiary Wastewater Treatment
- Tertiary Waste Water Treatment Methods
- Common Tertiary Wastewater Treatment Methods
- Reverse Osmosis -Tertiary Wastewater Treatment
- Electrodialysis – Tertiary Wastewater Treatment
- Filtration
- Conclusion
What is Tertiary Wastewater Treatment
Tertiary wastewater treatment is the final and most advanced stage of the purification process. It is designed to remove contaminants that primary and secondary treatment cannot eliminate. It focuses on polishing the effluent by targeting fine suspended solids, nutrients like nitrogen and phosphorus, dissolved pollutants, and harmful microorganisms. By using filtration, chemical dosing, membrane processes, and disinfection, tertiary systems ensure the water meets strict environmental and reuse standards. Understanding what is tertiary wastewater treatment is essential because it plays a key role in protecting ecosystems, supporting industrial reuse, and ensuring a safer, cleaner water supply for communities.

Tertiary treatment of wastewater makes it ready for reuse. Some examples of reuse include:
- Reclaimed water finds use in cooling systems, boiler feed, process water, and other industrial applications.
- Reuse in agriculture, horticulture, lawn watering, golf courses, and other applications
- Finds use in groundwater recharge to supplement groundwater resources for downstream consumers or to keep saline water out of coastal areas.
Tertiary Waste Water Treatment Methods
Tertiary wastewater treatment methods provide advanced purification beyond primary and secondary stages, ensuring high-quality effluent suitable for discharge or reuse. These methods remove fine solids, nutrients, pathogens, and dissolved contaminants using specialized physical, chemical, and membrane-based processes designed for maximum water clarity and safety.
Physicochemical methods use physical actions and chemical reactions to remove pollutants that biological treatment cannot fully eliminate, making them advanced wastewater treatment processes. These methods target fine suspended particles, dissolved solids, nutrients, heavy metals, and microorganisms by enhancing separation, adsorption, or chemical transformation. Physicochemical methods deliver precise and highly effective purification in tertiary wastewater treatment, producing high-quality effluent suitable for discharge or reuse.
Common Tertiary Wastewater Treatment Methods
Here are the common physicochemical methods used in tertiary wastewater treatment:
- Filtration (Sand, Dual-Media, Multimedia)
- Membrane Filtration (UF, NF, RO)
- Activated Carbon Adsorption
- Ion Exchange
- Chemical Coagulation and Flocculation
- Disinfection (Chlorination, UV, Ozone)
- Nutrient Removal (Phosphorus & Nitrogen Removal)
- Advanced Oxidation Processes (AOPs)
- Constructed Wetlands
- Electrodialysis
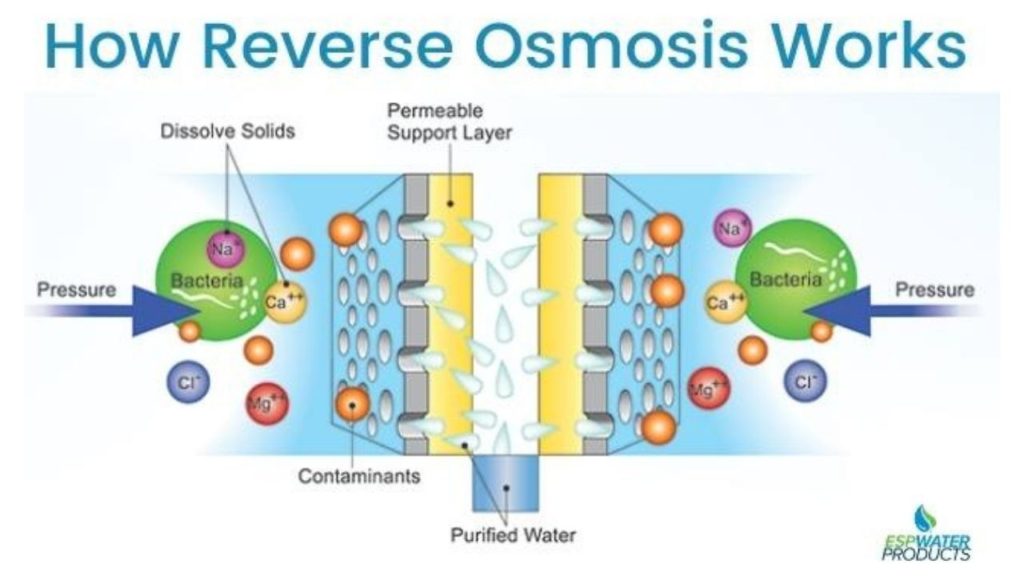
Reverse Osmosis -Tertiary Wastewater Treatment
Reverse Osmosis produces demineralized water by forcing water through semipermeable membranes at high pressure. We apply a pressure greater than the osmotic pressure across a membrane that separates a concentrated solution and a dilute phase.

This drives the solvent or water toward the dilute phase, making solute or impurity concentration rise on one side while pure water moves through the membrane. In Tertiary Wastewater Treatment, this method is widely used because it ensures high-quality purification. The Reverse Osmosis process requires pressures of about 4000 to 7000 kN/m² to achieve sufficient solvent flux.
The most crucial element is the permeable membrane, typically made from cellulose acetate, formamide, and magnesium perchlorate. These membranes need large surface areas for effective treatment. To optimize space, membrane modules are preferred over single sheets. This is especially true in advanced systems used for Tertiary Wastewater Treatment.
Reverse osmosis finds applications in the following:
- Separation of toxic ions from plating wastes.
- Desalting seawater to produce drinking water.
- Concentration of radioactive wastes.
- Removal of organics from vegetable and animal wastes.
Electrodialysis – Tertiary Wastewater Treatment
Electrodialysis is another popular tertiary wastewater treatment method that employs the removal of the solute from the solution instead of removing the solvent. This process uses selectively permeable membranes and an electric potential difference to separate ions from a solution. The electric power required depends on the number of ions removed from the water.
An electrodialysis cell contains anionic and cationic membranes arranged alternately, creating multiple compartments between electrodes placed at each end. When an electric voltage is applied, anions migrate toward the positive electrode and cations move toward the negative electrode. As a result, solutions in alternating compartments become dilute while others become more concentrated. Once the process reaches the desired degree of separation, the system removes the solutions. This selective ion removal makes electrodialysis an effective option in tertiary wastewater treatment, especially when precise control over dissolved salts is needed.
Filtration
Tertiary treatment removes total suspended solids (TSS). It eliminates the fine particles left after the secondary clarifying process. Proper pretreatment is essential before filtration. The concentration of suspended particles in the influent must be below 100 mg/l. This ensures efficient filtration.
The most common filtration methods include diatomaceous earth filtration and pressure filtration. Sand filtration is used with standard and multimedia units. Ultrafiltration and the moving-bed filter are also common. All these systems work by physically straining finely separated particles. Filtration is a key step in tertiary wastewater treatment.
Diatomaceous Earth Filtration
Diatomaceous earth filtration is a type of mechanical separation that involves filtering wastewater with diatomaceous earth, a powdered filter aid, on a supporting media. As the filtration progresses, the solid material that will not pass through the diatomaceous earth accumulates on the filter. Eventually, this builds up pressure that prevents filtration. After that, we backwash the filter and remove the accumulated material to prepare it for the next round of filtration.
Sand Filtration
The standard method of filtration uses sandbeds with graded sand. The sand is placed on a supporting medium. An underdrain collects the filtered effluent. Solids will build up and eventually block the holes as wastewater containing solids passes through this type of filter. This results in excessive head loss and/or poor effluent quality. As a result, sand filtration demands some provisions for the removal of the accumulated material. Backwashing the sand, or reversing the flow with air scour, helps to keep the sand in suspension while washing away the lighter material.
Ultrafiltration
Ultrafiltration (UF) is a widely used tertiary wastewater treatment method that purifies water by forcing it through a semipermeable membrane. In this process, water and low-molecular-weight solutes pass to the permeate side, while suspended particles and larger solutes stay on the retentate side. As one of the most reliable physicochemical treatment methods, UF removes most organic compounds, viruses, and many dissolved salts. People highly prefer it because it delivers consistent water quality regardless of the source, occupies minimal space, and eliminates 90–100% of pathogens. Moreover, ultrafiltration supports advanced purification without chemicals, except during membrane cleaning. Compared to other types of tertiary wastewater treatment, UF operates at much lower pressures—around 50 psi—while reverse osmosis often requires pressures above 500 psi, making UF energy-efficient and cost-effective.

Shall we wrap up?
Key Takeaways
- Tertiary Wastewater Treatment is the final stage of purification, targeting contaminants that earlier processes cannot remove.
- It uses methods like filtration, chemical dosing, disinfection, and membrane processes to ensure high-quality effluent.
- Common techniques include sand filtration, reverse osmosis, and ultrafiltration, which enhance water treatment efficacy.
- This stage supports water reuse in agriculture, industry, and groundwater recharge, protecting ecosystems and public health.
- Understanding Tertiary Wastewater Treatment is crucial for achieving environmental compliance and safe water supplies.
Conclusion
Tertiary wastewater treatment plays a crucial role in producing high-quality effluent suitable for discharge or reuse. In this blog, we briefly explored advanced methods such as reverse osmosis, electrodialysis, ultrafiltration, and other physicochemical techniques that provide superior purification compared to primary and secondary stages. Each method has its own advantages, and depending on the final application—whether irrigation, industrial reuse, or safe release into natural water bodies—engineers may choose a single technique or a combination of multiple processes. These tertiary wastewater treatment methods help remove fine solids, nutrients, pathogens, and dissolved contaminants, ensuring strict environmental standards are met. Ultimately, tertiary treatment strengthens water sustainability by delivering cleaner, safer, and more reliable treated water for diverse needs.



