The activated sludge process is a widely used and highly effective method in wastewater treatment, employing aerobic microorganisms to remove organic pollutants efficiently. This guide covers the activated sludge process definition, stages, and diagram, while explaining its role in secondary wastewater treatment for producing high-quality effluent. You will understand how aeration tanks, wastewater treatment microorganisms, mixed liquor suspended solids (MLSS), and return activated sludge (RAS) work together to maintain stable and efficient system performance. The activated sludge process in wastewater treatment relies on proper aeration, controlled sludge recycling, and effective process monitoring. Due to its reliability, adaptability, and high treatment efficiency, activated sludge wastewater treatment remains the preferred choice for many municipal and industrial wastewater treatment plants, ensuring consistent performance and environmental compliance.
Activated Sludge Process is a globally used wastewater treatment technique. In the previous blogs, I had shown you the various secondary wastewater treatment techniques. In this blog, we will dig deep into this widely used technique of activated sludge process, its configuration, process control and aeration methods.
Activated sludge Process – Definition
The activated sludge process is a type of wastewater treatment that uses aeration. It involves a biological floc made up of bacteria and protozoa to clean sewage or industrial waste waters. This process is biological and has various applications. It includes oxidising carbonaceous biological matter. Additionally, it deals with nitrogenous waste in the biological matter, mostly ammonium and nitrogen.
Before starting make sure that you take a quick glance through the blog, Wastewater Treatment- Stages and Process full details for better understanding

The activated sludge process employs aerobic microorganisms that can digest organic substances in sewage. Also, they have the ability to cluster together via flocculation. The flocculated particles settle out as sludge. As a result, the liquid coming out is relatively free of suspended solids and organic matter.
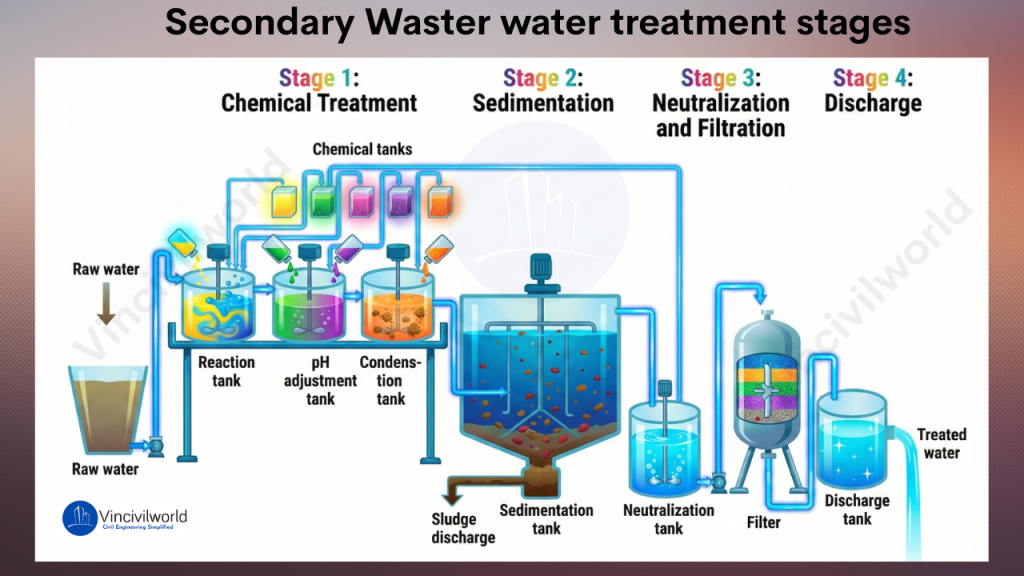
Activated Sludge Process – Stages, Configuration, and Diagram
The activated sludge process is a widely used secondary wastewater treatment method. It removes carbonaceous organic pollution through aerobic biological activity. The configuration of an activated sludge wastewater treatment system consists of interconnected units that operate together to ensure efficient treatment.
Aeration Tank
The aeration tank is the heart of the activated sludge process in wastewater treatment. Primary treated sewage is mixed with microorganisms, forming mixed liquor suspended solids (MLSS). Air or oxygen is supplied using diffusers or mechanical aerators to support aerobic microbes. These microorganisms consume organic matter and convert it into stable end products.
Secondary Settling Tank (Secondary Clarifier)
The mixed liquor flows into the secondary settling tank, where biological flocs settle by gravity. Clarified effluent overflows for further treatment or discharge. Settled sludge collects at the bottom.
Sludge Recycling System
A portion of the settled sludge is returned as Return Activated Sludge (RAS) to maintain microbial concentration in the aeration tank. Excess sludge is removed as Waste Activated Sludge (WAS) to control sludge age and system stability.
Advanced control systems regulate aeration, sludge recycling, and flow rates, ensuring stable and efficient secondary wastewater treatment performance.
Now, how about looking deep into what happens inside an activated sludge process?

Activated Sludge Process Steps
The activated sludge process operates through a series of well-defined stages that enable effective secondary wastewater treatment. Each stage supports biological activity, sludge separation, and recycling to achieve high organic pollutant removal and regulatory compliance.
- After primary treatment, wastewater enters into an aeration tank. A portion of sludge from the secondary settling tank also enters.
- Organic matter comes into close contact with sludge from the secondary settling tank. Sludge is densely populated with microorganisms that are actively growing.
- Diffusers or surface aerators inject air in the form of bubbles into the sewage-sludge mixture.
- Microorganisms break down organic matter into stable chemicals like NO3, SO4, and CO2 while also producing new bacterial cells.
- The effluent along with the actively growing microbial population passes to the secondary settling tank.
- The secondary settling tank separates the aeration tank’s effluent, which contains flocculent microbial matter into supernatant and sludge. The treated supernatant undergoes further treatment before discharge.
- This sludge from the settled waste returns to the aeration system’s inlet to re-seed the new wastewater reaching the tank. Return activated sludge (R.A.S.) is the fraction of the floc that returns to aeration tank.
- The remaining sludge goes to sludge digesters for further treatment and safe disposal.
“Mixed liquor” refers to the combination of the liquid and microorganisms in the aeration tank. The suspended solids are called “Mixed Liquor Suspended Solids” (MLSS).
In the next section, we will find out the basic process control parameters in an activated sludge process.

Process Control in Activated Sludge Process
The general process control method monitors the following variables:
- Sludge Volume Index (SVI)
- Mean Cell Residence Time (MCRT)
- Food to Microorganism Ratio (F/M)
- Dissolved oxygen (DO)
- Biochemical oxygen demand (BOD)
- Chemical oxygen demand (COD)
Let me explain these parameters in detail.
Sludge Volume Index
Sludge Volume Index measures the volume of settled sludge in milliliters. This volume is occupied by 1g of dry sludge solids after 30 minutes of settling in a 1000 milliliter graduated cylinder. It gives a measure of the settling ability of the sludge. SVI ranges from 40 to 100 for a good sludge which settles down easily. Bulking Sludge is a biomass consisting of filamentous organisms with very poor settling characteristics. For a bulking sludge, SVI value can exceed 200. Sufficient pH control, adequate aeration and addition of hydrogen peroxide to the aeration tank prevents bulking.
Mean Cell Residence Time
Mean Cell Residence Time is the ratio of total mass (lbs) of mixed liquor suspended solids in the aerator and clarifier to the mass flow rate (lbs/day) of mixed liquor suspended solids leaving as final effluent.
Food to Microorganism Ratio
The Food to Microorganism Ratio indicates how much organic matter is fed to the microorganisms each day. It is relative to the mass of microorganisms under aeration. In other words, it is the ratio of the amount of BOD fed to the aerator (lbs/day). It is compared with the amount (lbs) of Mixed Liquor Volatile Suspended Solids (MLVSS) under aeration.
Main Control Parameters
The mean cell residence time and F/M Ratio are the main control parameters used industrially. Both are directly related to the effluent quality. However, it is tedious to control the plant on the basis of the F/M ratio since it necessitates a lot of laboratory work to find the BOD and MLSS in the system. Therefore, the mean cell residence time is the best choice for controlling an activated sludge system.
Now you got an idea about the entire process and its important parameters. Next, we move on to the various aeration methods.
Aeration Methods in Activated Sludge Process
The decomposition of organic waste requires a very high concentration of oxygen at the initial stages of contact between microorganisms and the organic matter. The conventional systems usually maintain a plug flow hydraulic regime and keeps aeration and a mixing at an uniform rate along the entire tank. As a result, the oxygen concentration drops rapidly in the inlet and this can harm the microbes.
At the outlet, there is a surplus of oxygen which is not necessary and leads to economical losses. In order to match the oxygen supply and demand along the entire journey of wastewater from inlet to outlet, the mode of aeration needs some modifications. Let’s have a look at the different aeration methods in an activated sludge process.
Diffused Aeration
Sewage liquor is pumped into large tanks with floor-mounted diffuser grid aeration devices. Passing air creates a curtain of bubbles that oxygenates the liquor while also mixing it. An air blower usually creates the air. Oxygen replaces air for unusually strong and difficult to treat sewage.
Tapered Aeration
The organic waste needs more oxygen at the inlet. As it degrades progressively its oxygen demand decreases. Tapered aeration works on this principle. Aeration is intense at the inlet and decreases progressively along the length of the aeration tank. As this method involves the more efficient use of air, it results in savings in the pumping costs too.
Step Aeration
This method aims to equalize the oxygen supply and its demand. It introduces fresh feed at several points in the aeration tank, while keeping the rate of oxygen supply constant. This ensures a more even oxygen distribution over the entire tank and throughout the aeration stage. Baffles divide the aeration tank into several channels with each channel representing one step of the process.
Complete Mix Activated Sludge Process
In complete mix process, the aeration tank receives a mixture of fresh feed and recycled sludge at several locations within the tank. This ensures a constant supply and demand of oxygen along the length of the tank.
Contact Stabilisation
The microbial mass comes in contact with wastewater for short durations of time, approximately 0.5 to 1 hour in the biosorption unit. An anaerobic digestion unit stabilizes the resulting sludge after a retention period of about 2-3 hours. In the digestion unit, microbes consume the organic wastes removed in the biosorption unit. Since we stabilize the return sludge with higher solid concentrations, this reduces the volume of the aeration tank.
Pure Oxygen Activated Sludge Process
This type of activated sludge process supplies pure oxygen instead of air. It recirculates this oxygen into well mixed and converted chambers. Conventional processes utilize only 5-10% of oxygen. In contrast, the pure oxygen activated sludge process ensures about 90% utilization of oxygen. Further, it results in higher bacterial activity, lower sludge volume and sludge with better settling characteristics.
That’s it about activated sludge process. Let us know in the comments if you wish to know more.
Key Takeaways
- The activated sludge process effectively treats wastewater using aerobic microorganisms to remove organic pollutants.
- Key stages include aeration tanks, secondary settling tanks, and sludge recycling to maintain treatment efficiency.
- Proper aeration and control parameters like SVI and F/M Ratio are essential for optimal performance.
- Various aeration methods, such as diffused and tapered aeration, improve oxygen supply and microbial activity.
- This process provides a reliable solution for secondary wastewater treatment, ensuring cleaner effluent.
Conclusion
The activated sludge process is a proven method for secondary wastewater treatment. It is highly efficient and used worldwide. This process removes organic pollutants from sewage and industrial wastewater. This process promotes aerobic microbial activity in aeration tanks. It achieves effective BOD reduction, suspended solids removal, and stable effluent quality. Proper control of operational parameters, such as dissolved oxygen, MLSS, and sludge age, ensures consistent system performance. It also prevents common issues like sludge bulking. With multiple activated sludge process configurations and aeration methods, the system can adapt to varying wastewater loads and treatment requirements. Due to its flexibility, reliability, and high treatment efficiency, the activated sludge process remains a cornerstone of modern biological wastewater treatment systems and sustainable water management.